Proteins and carbohydrates are important constituents of food. The best long-term solution to disposal would be to recycle polymer waste. Burning them isn’t good because poisonous gases form as a result. They are a major source of visual pollution and fill up available waste sites. Polymers are non-biodegradable, meaning that they do not decay. Here is the official structure of tetrylene as drawn in the CIE syllabus: These monomers are held via an ester link as shown below: In polyesters (such as tetrylene) the two monomers are a diacid and a diol. Here is the official structure of nylon as drawn in the CIE syllabus: These two monomers join together via an amide link as shown below: In polyamides (such as nylon) the two monomers are always a dicarboxylic acid (or diacid) and a diamine. During the reaction a water molecule is lost in the process (therefore condensation).  
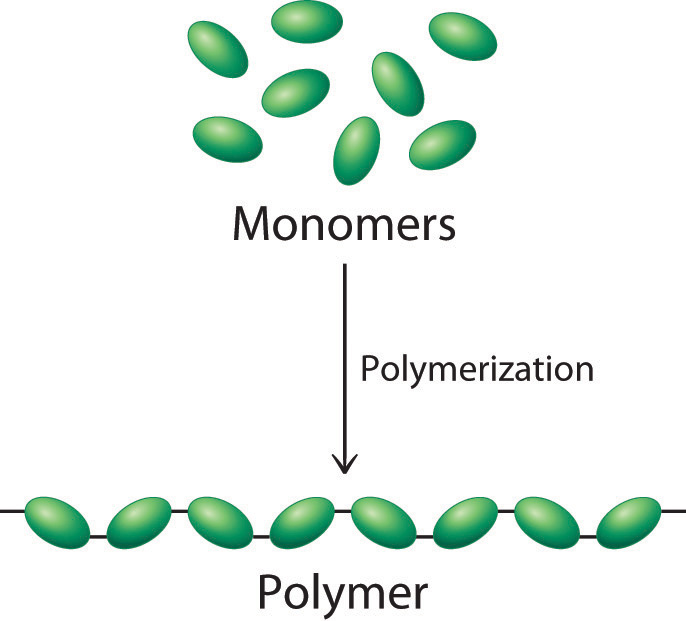
In a condensation reaction, two monomers react together and join. Take notice of the fact that the double bond has become a single bond in the polymer. The contents inside the square brackets indicate the repeating unit (monomer) with the letter ‘n’ indicating the total number of the monomers throughout the structure. This have been covered briefly in the topic alkenes.īy breaking apart the double bond of alkenes, repeating units (monomers) can be joined together into a larger, longer molecule (polymer). There are two main methods of polymerization:īoth nylon and tetrylene are made via condensation polymerization. These two polymers will be covered in more detail down below Synthetic polymers are man-made polymers such as nylon (polyamide) and terylene (polyester). Different polymers are built from different monomers and have varying linkages between the monomers. Dehydration reactions typically require an investment of energy for new bond formation, while hydrolysis reactions typically release energy by breaking bonds.Polymers are large molecules built from small units (monomers). Dehydration and hydrolysis reactions are similar for all macromolecules, but each monomer and polymer reaction is specific to its class. When polymers are broken down into smaller units (monomers), a molecule of water is used for each bond broken by these reactions such reactions are known as hydrolysis reactions. These types of reactions are known as dehydration or condensation reactions. A monomer joins with another monomer with the release of a water molecule, leading to the formation of a covalent bond. 
The polymer is more than the sum of its parts: it acquires new characteristics, and leads to an osmotic pressure that is much lower than that formed by its ingredients this is an important advantage in the maintenance of cellular osmotic conditions. Macromolecules are made up of single units known as monomers that are joined by covalent bonds to form larger polymers. Proteins, carbohydrates, nucleic acids, and lipids are the four major classes of biological macromolecules-large molecules necessary for life that are built from smaller organic molecules. Breakdown of these macromolecules provides energy for cellular activities. Proteins are broken down by the enzymes pepsin and peptidase, and by hydrochloric acid. For instance, carbohydrates are broken down by amylase, sucrase, lactase, or maltase. Each macromolecule is broken down by a specific enzyme. This allows for easy absorption of nutrients by cells in the intestine. For example, in our bodies, food is hydrolyzed, or broken down, into smaller molecules by catalytic enzymes in the digestive system. These reactions are similar for most macromolecules, but each monomer and polymer reaction is specific for its class. \).ĭehydration and hydrolysis reactions are catalyzed, or “sped up,” by specific enzymes dehydration reactions involve the formation of new bonds, requiring energy, while hydrolysis reactions break bonds and release energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
